Problem 1.14:
Solution:
Given: \( \gamma=1.3 \)
\( A_{1} = A_{e}=A \),
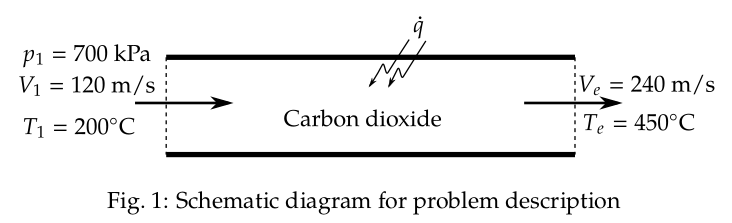
\( V_{1} = 120\,\text{m/s} \),
\( T_{1} = 200^{\circ}\,\text{C} = 473\,\text{K} \),
\( p_{1} = 700\,\text{kPa} \),
\( V_{e} = 240\,\text{m/s} \),
\( T_{e} = 450^{\circ}\,\text{C} = 723\,\text{K} \).
To calculate: \( \dot{q}/\dot{m}=? \), \( \dot{m}/A=? \).
The schematic diagram of the problem description is shown in Fig. 1.
Using the molar mass of carbon-dioxide as 44 kg/kmol, and the universal gas constant as \( 8314\,\text{J/kmol-K} \), the gas constant can be written as,
The heat capacity at constant pressure can be calculated as,
--- Ad ---
---
Applying the conservation of energy principle,
$$ c_{p}T_{1}+\frac{V_{1}^{2}}{2}+\dot{q}/\dot{m}=c_{p}T_{e}+\frac{V_{e}^{2}}{2} $$We can solve for the heat added per unit mass to be,
--- Ad ---
---
The density at the inlet can be calculated using the ideal gas equation to be,
The mass flow rate,
$$ \dot{m}=\rho_{1}A_{1}V_{1} $$which gives the mass flow rate per unit area as,