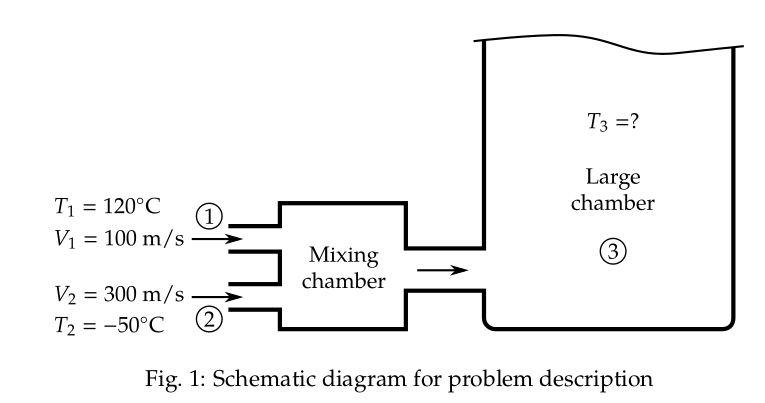
Problem 1.5:
Two jets of air, each having the same mass flow rate, are thoroughly mixed and then discharged into a large
chamber. One jet has a temperature of
oC
and a velocity of
m/s,
whereas the other has a
temperature of
oC
and a velocity of
m/s.
Assuming that the process is steady and
adiabatic, find the temperature of the air in the large chamber.
Solution:
A schematic diagram of the configuration is shown in Fig. 1.

Given:
The mass flow rate is same through the two inlets, therefore,
$$ \dot{m}_{1}=\dot{m}_{2}=\dot{m} $$ $$ \dot{m}_{3}=\dot{m}_{1}+\dot{m}_{2}=2\,\dot{m} $$--- Ad ---
---
Applying the conservation of energy equation (without heat and work),
$$ \left(\dot{m}_{1}\,c_{p}\,T_{1}+\dot{m}_{1}\frac{V_{1}^{2}}{2}\right) + \left(\dot{m}_{2}\,c_{p}\,T_{2}+\dot{m}_{2}\frac{V_{2}^{2}}{2}\right) = \left(\dot{m}_{3}\,c_{p}\,T_{3}+\dot{m}_{3}\frac{V_{3}^{2}}{2}\right) $$ $$ \left(\dot{m}\,c_{p}\,T_{1}+\dot{m}\frac{V_{1}^{2}}{2}\right) + \left(\dot{m}\,c_{p}\,T_{2}+\dot{m}\frac{V_{2}^{2}}{2}\right) = \left(2\,\dot{m}\,c_{p}\,T_{3}+2\,\dot{m}\frac{V_{3}^{2}}{2}\right) $$ $$ \left(c_{p}\,T_{1}+\frac{V_{1}^{2}}{2}\right) + \left(c_{p}\,T_{2}+\frac{V_{2}^{2}}{2}\right) = \left(2\,c_{p}\,T_{3}+2\frac{V_{3}^{2}}{2}\right) $$ $$ T_{3}=\frac{c_{p}\,T_{1}+\frac{V_{1}^{2}}{2} + c_{p}\,T_{2}+\frac{V_{2}^{2}}{2} - V_{3}^{2}}{2\,c_{p}} $$Assuming \( c_{p}=1005\,\text{J/kg-K} \) for air and the velocity in the large chamber to be effectively zero,